Since this chapter only considers the p-block elements, for the purposes of this chapter this system is functionally identical to system 2. This definition adds Zn, Cd, and Hg (and sometimes Cu, Ag, and Au) since they form ions with an (n-1) d 10 valence electron configuration such as Zn 2 +, Cd 2 + and Hg 2 +(and Cu +, Ag +, and Au +). Metals which follow the transition elements in the sense of forming ions with a completely full (n-1)d valence shell sometimes along with Al and the p-block metalloids.However, it has the disadvantage of excluding metals like Zn, Cd, and Hg, which form many compounds in which the metal has a (n-1) d 10 configuration. Consequently, it will be used here in the sections which follow. This system has the advantage of emphasizing the interesting and unique properties of the metals and metalloids of p-block as well as continuities in those properties through the p-block. Another disadvantage of this system is that it entangles the issue of which elements should be classified as post-transition metals with the thorny issue of which elements should be classified as metals vs. However, if this scheme is adopted too rigidly Al is excluded since it technically doesn't follow the d-block (and has an unfilled (n-1) d subshell) and the metalloids are excluded, even though many of them also form relatively soft and electron rich cations with filled (n-1) d subshells. By this definition only the metals in groups 13 and higher and Rows 3 and higher which form relatively soft and electron rich cations and exhibit significant covalency in their bonding are included. Several systems are used to classify elements as belonging to the post transition metals. Their electron richness means that they tend to form soft cations. Roughly, this translates into relatively lower melting points (since more antibonding levels in the band structure tend to be occupied), increased preference for covalency, and greater brittleness than other metals (due to the resulting directional bonding). The main features of the post transition metals are that they are relatively electron rich and electronegative compared to what is classically thought of as a metal. Fortunately, in practice it is less important to precisely define what is and is not a post transition metal than to understand the reason why it might be helpful to classify an element as a post-transition metal. As with the metalloid concept there is no universal consensus as to what exactly is a post transition metal. The term post-transition metals refers to those elements that are metals following the transition metals. The group 13 elements include post-transition metals. Gallium (Ga), Indium (In), Thallium (Tl), and Nihonium (Nh, marked under its old symbol, Uut, in the Figures on this page), post transition metals that exhibit the inert pair effect to varying degrees.Aluminum, Al, an electron deficient 3rd row element sometimes classified as a metal and sometimes as a metalloid.Boron, B, an electron deficient 2nd row element sometimes classified as a nonmetal and occasionally as a metalloid. 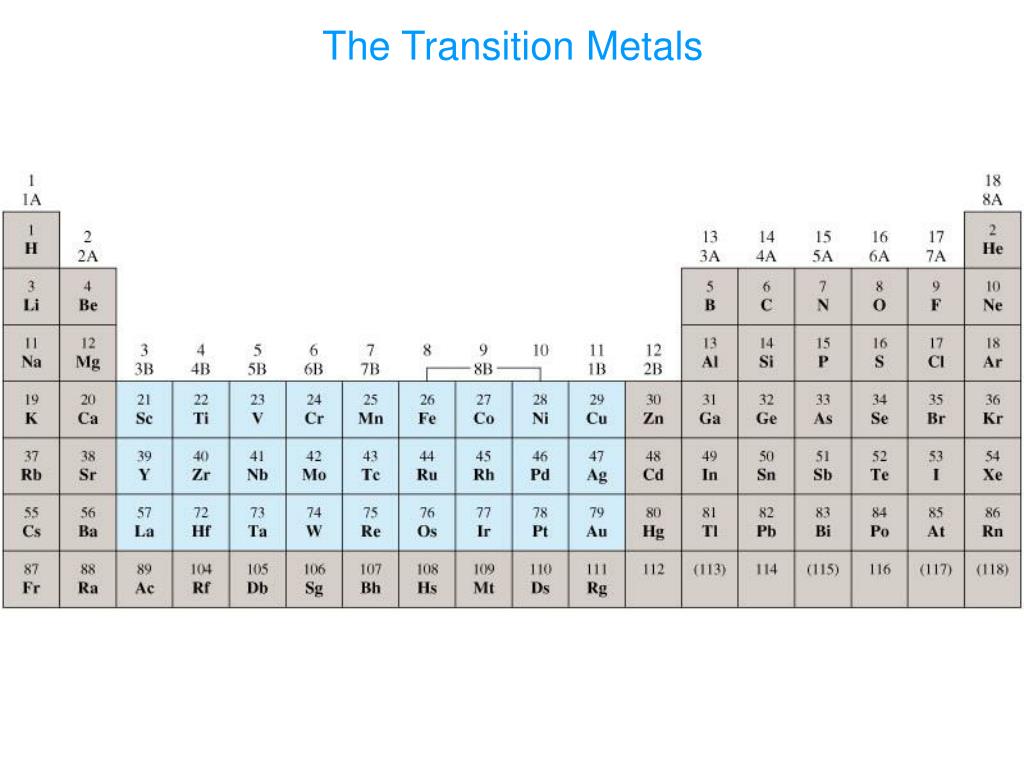
The group 13 elements are chemically diverse, comprising elements: 
Adapted from the periodic table at https :///Bookshelves/General_Chemistry/Map%3A_Chemistry_-_The_Central_Science_(Brown_et_al.)/02._Atoms%2C_Molecules%2C_and_Ions/2.5%3A_The_Periodic_Table Note that since the periodic table used was developed, element 113 was named Nihonium and given the symbol Nh. Position of the group 13 elements in the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
